IEST's Testing Solutions in the Field of Sodium-ion Batteries
Compared with lithium-ion batteries, sodium-ion batteries have lower energy density, but have better battery safety performance, high and low temperature performance, and rate performance. They are expected to become an important supplement in the field of energy storage batteries and two-wheeled electric vehicles. It is expected that by 2025, sodium-ion battery production capacity will exceed 25GWh.
After years of industrial exploration, current sodium-ion battery cathode materials with high feasibility for mass production are divided into three routes: layered metal oxide systems, polyanion ionic compound systems and Prussian-type compound systems, these three technical routes each have their own advantages and disadvantages. The structure of layered metal oxides is similar to that of lithium-ion ternary cathode materials. It has a relatively high specific capacity and good comprehensive performance, by adjusting the selection and proportion of transition metal elements (including Ni, Co, Mn, Ti, V, Cr, Fe, Cu, etc.), it can take into account the needs of multiple scenarios such as power and energy storage and is the preferred solution for recent industrialization.
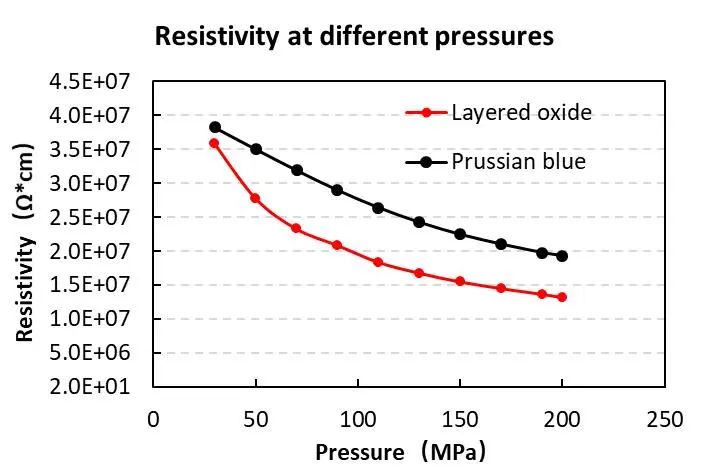
Figure 1. Resistivity test results of Prussian blue and layered metal oxide powder
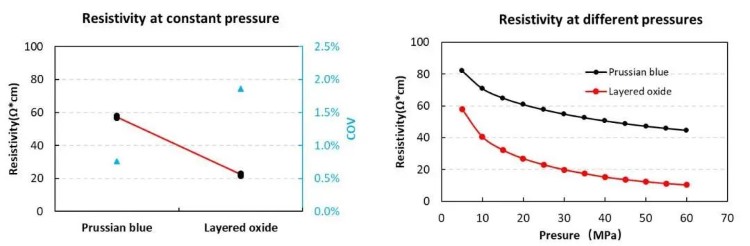
On the sodium-ion battery cathode material, IEST used a powder resistance and compaction density meter (PRCD3100) to verify the resistivity relationship between layered oxide and Prussian blue at the powder level. The results are shown in Figure 1. After the two powders were prepared into pole pieces, the resistivity relationship at the pole piece level was verified using a pole piece resistance meter (BER2300). The results are shown in Figure 2. We can find that the resistivity of Prussian blue is greater than the layered oxide at the powder level; at the pole piece level, the Prussian blue resistivity is greater than the layered oxide resistivity. The resistivities of the powder and pole pieces are the same in law.
Figure 2. Resistance test results of Prussian blue compounds and layered metal oxide electrodes
2