In-situ Gas Production Analysis of Cell Overcharge—Electrolyte Additives
Gas production in lithium-ion cells is an important cause of fire and explosion of cells,the reasonable selection and matching of the four main materials(Cathode material, Negative electrode material, Electrolyte, Separator) will also affect the safety performance of the cell. The Figure 1 is the gas production behavior of different electrolyte 1. Under different potentials, the cells will have different degrees of gas production reaction. Electrolyte is an important channel for the transmission of lithium ions, and its electrochemical activity plays a key role in the dynamics and thermodynamic properties of the cell. During the overcharging process of the cell, the additives in the electrolyte are likely to undergo an oxidation reaction, and a certain amount of gas is generated to cause the cell to swell2-4. In this paper, an In-Situ Gassing Volume Analyzer(GVM) was used to perform in-situ overcharge volume test on NCM523/graphite cells (theoretical capacity 1000mAh) with different additive types and contents, and analyze the gas production behavior of the cells.
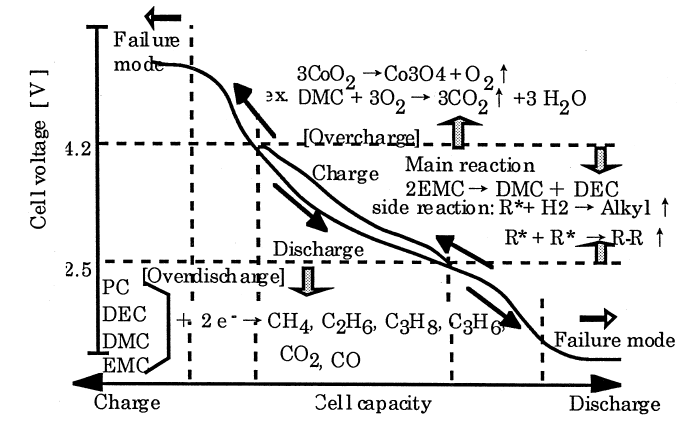
Figure 1. The Gas production behavior of different electrolyte1
1.Experimental Equipment and Test Methods
1.1 Experimental Equipment: Model GVM2200(IEST),test temperature range 20~85℃,support dual-channel (2 cells) synchronous test,the appearance of the equipment is shown in Figure 2.

Figure 2. The Appearance of GVM2200
1.2. Test Parameters: 25℃ 1C CC to 5V.
1.3. Test Method: Initially weigh the cell m0, put the cell to be tested into the corresponding channel of the device, open the MISG software, set the cell number and sampling frequency parameters corresponding to each channel, the software automatically reads the volume change, test temperature, current, voltage , capacity and other data.
2.Gas Production Analysis of Cell Overcharge In-situ
2.1 Charge-discharge Curve and Volume Change Curve Analysis
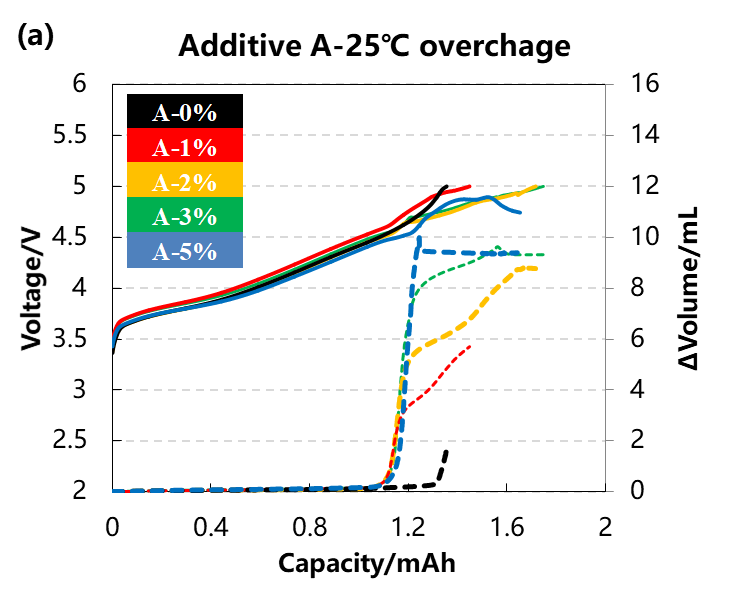
The volume change curve and voltage curve of the cell are shown in Figure 3(a)(b),The content of the two additive types A and B:0%、1%、2%、3%、5%. It can be seen from the curve that with the increase of the additive content, the volume changes of the two groups of cells are getting larger, indicating that the gas production reaction of the additive causes the cell swelling. When the content of additives reaches 5%, it can be seen from the voltage curve that it is difficult for the voltage of the cell to reach the upper limit of 5V, this may be due to the fact that the cell produces more gas, which leads to the deterioration of the interface contact between the electrodes and the greater polarization of the cell.
 |
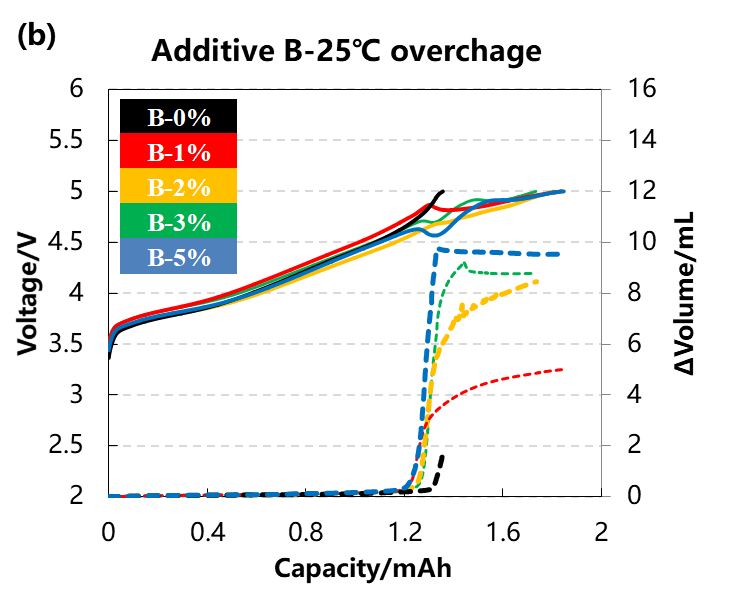 |
Figure 3. (a)The charge and discharge and volume change curve in different content of additive A. (b)The charge and discharge and volume change curve in different content of additive B.
2.2 Analysis of Gas Production Capacity and Inflection Voltage of Cells
The cell charging capacity and the inflection point voltage information of the gas production curve with different types and contents of additives are shown in Table 1 and Figure 4. With the increase of the additive content, the total gas production of the cells corresponding to the A and B additive types are gradually increasing, and the gas production corresponding to A is slightly more than that of B. The onset voltage of gas production gradually decreases with the increase of the additive content, and the voltage corresponding to A is lower than that of B. This shows that the gas production potential of A is earlier than that of B, show if A is used as an anti-overcharge protection additive, overcharge can be determined earlier, and corresponding safety protection measures can be taken in time.
Table 1. Cell charging capacity and gas production related information corresponding to different additive types and contents

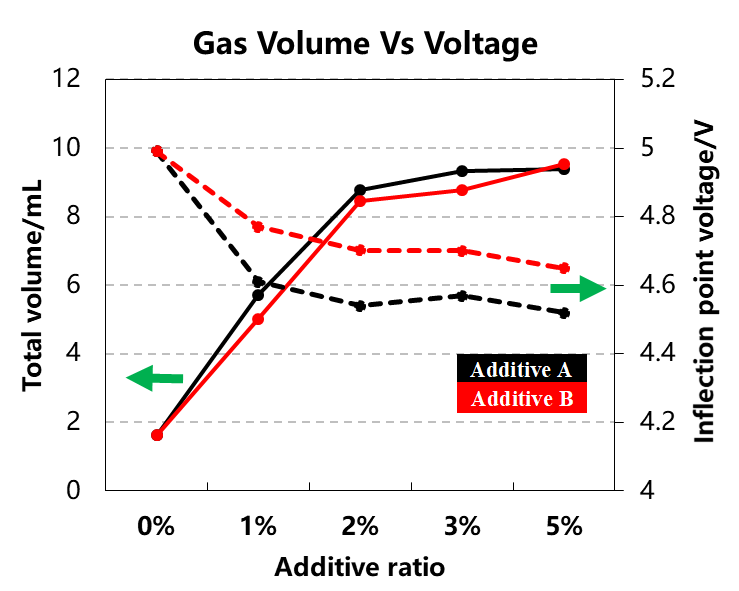
Figure 4. Gas production volume and inflection voltage curve with different additive content
3.Summary
In this paper, a temperature-controllable dual-channel In-Situ Gassing Volume Analyzer is used to compare the overcharge gas production behavior of lithium-ion cells with two different additive types and contents. It can be found that the A additive has a lower reaction potential than the B additive, the total gas production is slightly larger, which can be better used as an anti-overcharge protection additive.
4.References
1. Kazuma Kumai, Hajime Miyashiro. Gas generation mechanism due to electrolyte decomposition in commercial lithium-ion cell. Journal of Power Sources. 81–82(1999):715–719.
2. Christoph R. Birkl, Matthew R. Roberts, Euan McTurk, Peter G. Bruce, David A. Howey. Degradation diagnostics for lithium ion cells. Journal of Power Sources. 341 (2017): 373-386.
3. C. P. Aiken, J. R. Dahn et al. An Apparatus for the Study of In Situ Gas Evolution in Li-Ion Pouch Cells. J. Electro Soc, 161(2014) A1548-A1554.
4. Randolph A. Leising. Abuse Testing of Lithium-Ion Batteries -Characterization of the Overcharge Reaction of LiCoO2ÕGraphite Cells. Journal of the Electrochemical Society, 148(8):A838-A844 (2001).
